|
2016, June: Rationale for continued study of Radioimmunotherapy
Comments on the Discontinuation of Bexxar
An advocates' perspective
The BEXXAR® therapeutic regimen (tositumomab and iodine I 131 tositumomab) WAS indicated for the treatment of patients with CD20-positive relapsed or refractory, low grade, follicular, or transformed non-Hodgkin's lymphoma who have progressed during or after rituximab therapy, including patients with rituximab-refractory non-Hodgkin's lymphoma.
 The sponsor cited the verh low usage as the reason for its decision to discontinue Bexxar. Why did this happen and what can we learn? Will this outcome have a chilling effect on the study of drugs that require something extra of oncologists to administer it? Could this bad outcome (for an effective drug) been avoided by the sponsor? The sponsor cited the verh low usage as the reason for its decision to discontinue Bexxar. Why did this happen and what can we learn? Will this outcome have a chilling effect on the study of drugs that require something extra of oncologists to administer it? Could this bad outcome (for an effective drug) been avoided by the sponsor?
In our view, the discontinuation of Bexxar shows that the accelerated approval of a drug is but a starting point - that the sponsor of the drug must also find its place in clinical practice by testing it head-to-head against regular treatments in different clinical settings.
... Consider, for example, that lacking such evidence that the decisions of a prescribing physician may be second-guessed when adverse events occur with the use of the newer agent - which are likely to occur in some patients with any approach.
While relatively very easy for the patient to receive, Bexxar is also a mildly "disruptive technology." It required more of oncologists to administer it - extra training and additional procedures. The administration of Bexxar was not especially difficult but it was apparently sufficiently burdensome to put off taking it on (or referring patients elsewhere) - without definitive evidence that the drug was better than regular treatments.
Controlled studies to test Bexxar against a standard protocol was left to the cooperative group (SWOG) testing CHOP-R against CHOP-Bexxar in high-risk advanced follicular lymphoma. http://clinicaltrials.gov/show/NCT00022945
... The SWOG study did not show a difference in outcomes at 4 years. The study asked a valid and important question, but the study question was not necessarily of primary interest to many patients ... who saw Bexxar as a way to potentially avoid the side effects of chemotherapy and its impacts on quality of life. This expectation is based on the Kaminski study and anecdotal accounts of very durable remissions (Our story among them).
... Further, in theory, pre-treatment with aggressive chemotherapy could work against the immune-mediated mechanism of Bexxar and it could also increase the risks -- to give one potent treatment shortly after another.
Why was a confirmatory study testing Bexxar as a single agent against regular treatment never done? The outcomes in the Kaminski study appeared to be the more compelling rationale for how to further test Bexxar: as a single agent against standard chemotherapy - perhaps most appropriately in patients without bulky disease. At the time, for example, CVP-R versus Bexxar in advanced, but non-bulky FL (< 8 cm?) in need to treat would have been a compelling study question.
To review outcomes for Bexxar as initial treatment for follicular lymphoma N Engl J Med. 2005 Feb, see Full Text
The discontinuation of Bexxar is a sad event for all of us for many reasons. While unproven, the unique features of Bexxar could make it more appropriate, or effective, or safer than the competing radioimmunotherapy in some clinical circumstances. About this we will never know.
The unique features of Bexxar radioimmunotherapy being:
* the shorter wavelength of the radiation and the longer half life;
* the differences in the absorbed radiation in normal tissue;
* the personalized dosing based on individual clearance rates detected by scans following the dosimetric dose.
* the use of the type II antibody that could be (in theory) more effective in patients resistant to Rituxan (a type I antibody).
See also Comparing Bexxar and Zevalin
In summary, I feel that sponsor has dropped the ball by failing to aggressively carry out the comparative studies needed to confirm the Kaminski findings. This has let patients down - in particular the participants of the clinical trials who took risks in order to make the approval of the drug possible. That ease of administration for the physician can be an important criteria for what is developed and adequately studied is cause for concern.
The above is old news I realize, but perhaps there are some lessons to be learned from the extinction of an effective drug that was easy to tolerate and over and done with in just a week. Recently I made comment during a Steering committee meeting that patients want to be free of the disease but also of the treatment - referring to a common approach for targeted therapies to give the drug indefinitely until relapse or unacceptable toxicity - which can also cause substantial financial toxicity.
..Indefinite use can be desirable I suppose depending on the daily impact on quality of life (will I feel good or off every day?) and you get a survival benefit in addition to a delay in relapse ... The desirable characteristic of Bexxar was that you were over and done with treatment in about a week , and there's a documented potential that the remission can last a decade or longer ... perhaps indefinitely in some persons.
KarlS
Send comment or question by
===
Bexxar™ is a monoclonal antibody that has a radioactive substance called iodine 131 attached to it. The monoclonal antibody in Bexxar seeks and binds to a protein receptor (named CD20) on the surface of both normal and malignant B cells. Once bound to the target cells, Bexxar delivers radiations, which enhances the killing effect of the antibody.
Normal b-cells will recover in 6 to 9 months because the parent b-cells do not have the CD20 receptor.
Technical name: Tositumomab, Iodine I 131 FDA.gov pdf
Who is Bexxar for? "BEXXAR is indicated for the treatment of patients with CD20 antigen-expressing relapsed or refractory, low grade, follicular, or transformed non-Hodgkin's lymphoma, including patients with Rituximab-refractory non-Hodgkin’s lymphoma. Determination of the effectiveness of the BEXXAR therapeutic regimen is based on overall response rates in patients whose disease is refractory to chemotherapy alone or to chemotherapy and Rituximab. The effects of the BEXXAR therapeutic regimen on survival are not known."
Source: Dailymed.nlm.nih.gov/ May 2008
Bulky disease might work against obtaining a complete response with Bexxar as with other therapies. For a discussion on how pretreatment with other agents might optimize Bexxar, see Bulky disease and RIT?
Note: Prior radiotherapy and extranodal disease does not seem to preclude use. "The BEXXAR therapeutic regimen should not be administered to patients with >25% lymphoma marrow involvement and/or impaired bone marrow 14 reserve (See WARNINGS and ADVERSE REACTIONS and Contraindications below)."
In the News
* Onc Times 2013:
Lymphoma Specialists Lament the Discontinuation of Underprescribed Bexxar http://bit.ly/16gKO10
* xconomy 2013:
Why Good Drugs Sometimes Fail: The Bexxar Story http://bit.ly/17hsYPO
And In the Pipeline 2013: Promise that didn't pan out. http://bit.ly/16KKmZ5
* Oncology Times: ONLINE FIRST:
Underprescribed Bexxar Discontinued by GlaxoSmithKline http://bit.ly/18yBbyc
How it works - mechanisms of action
Bexxar Mechanism-of-Action (MOA) from Jack Hyndman on Vimeo. When radio-labeled antibody binds to tumor cells it can cause tumor killing by:
(1) Self- killing (apoptosis) - programmed cell death triggered by the antibody
(2) Complement-dependent cytotoxicity (CDC) - where antibody fixes complement that kills the tumor cells
(3) Antibody-dependent cellular cytotoxicity (ADCC) - where effector cells (immune cells) kill the tumor cells
(4) Ionizing radiation from the radioisotope damages the tumor cells, leading to cell death
(5) Potentially, vaccine-like effect - leading to adaptive immunity against cells that survive initial treatment.
TECHNICAL
Clinical Pharmacology
"Utilizing the gamma emissions, relatively simple dosimetry can be performed and used to calculate the clearance rate of the radionuclide in an individual patient, thus determining the patient-specific therapeutic dose.
The half-life of I-131 (approximately 8 days) is also well suited for RIT [21], because it is similar to the half-life of murine antibodies in humans. Therefore, when I-131-labeled tositumomab is bound to CD20, a stable cell-surface molecule, the tumor receives high doses of radiation over several days.
Another advantage of I-131 is the relatively short average path length of the beta emissions (approximately 1 mm), which minimizes collateral damage to healthy tissue surrounding the tumor. As the I-131-conjugated antibody is metabolized, the free I-131 metabolites are released into the bloodstream and are rapidly excreted in the urine [22–24]. Free I-131 in the blood may also be taken up by the thyroid; however, accumulation in the thyroid can be effectively blocked by administering a supersaturated potassium iodine (SSKI) solution to patients before and during treatment, thus limiting potential damage. Nevertheless, thyroid dysfunction has been observed in approximately 5% of patients treated with Bexxar." Source:
Bexxar®: Novel Radioimmunotherapy for the Treatment of Low-Grade and Transformed Low-Grade Non-Hodgkin’s Lymphoma Julie M. Vose theoncologist
Imaging and Dosimetry
... The ultimate goal in drug therapy is to provide the most effective dose to optimize benefit and minimize toxicity. The tositumomab and 131I-tositumomab therapeutic regimen seeks to approach this goal by evaluating individual patient drug distribution and elimination over time.
These aspects of drug behavior are termed pharmacokinetics. With conventional pharmaceutical agents, the best that clinicians can do in this regard is to measure the time course for the drug (or metabolites) in the blood.
With radiolabeled agents, however, a much more accurate estimate of patient-specific pharmacokinetics is possible. Because 131I is both a 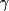 -emitter and a ß-emitter, it is possible to obtain whole-body -emitter and a ß-emitter, it is possible to obtain whole-body 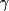 -counts from imaging of the patient after a relatively small (dosimetric) dose (184 MBq [5 mCi]) of 131I-tositumomab to estimate the total-body residence time (TBRT). This determination allows consideration of the factors shown to be influential in the pharmacokinetics of the tositumomab and 131I-tositumomab therapeutic regimen, such as extent of disease, bone marrow involvement, spleen size, and renal function (5). In addition to enabling an estimation of TBRT, imaging also provides an opportunity for the nuclear medicine physician to evaluate biodistribution with the same isotope, unlike other regimens that use a -counts from imaging of the patient after a relatively small (dosimetric) dose (184 MBq [5 mCi]) of 131I-tositumomab to estimate the total-body residence time (TBRT). This determination allows consideration of the factors shown to be influential in the pharmacokinetics of the tositumomab and 131I-tositumomab therapeutic regimen, such as extent of disease, bone marrow involvement, spleen size, and renal function (5). In addition to enabling an estimation of TBRT, imaging also provides an opportunity for the nuclear medicine physician to evaluate biodistribution with the same isotope, unlike other regimens that use a 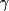 -emitting surrogate to predict biodistribution. -emitting surrogate to predict biodistribution.
However, unlike most nuclear medicine imaging procedures, this process is intended to evaluate gross biodistribution only, not provide detailed diagnostic information. The expected biodistribution after a dosimetric dose of 131I-tositumomab is illustrated by whole-body scans of a patient with the typical pattern for NHL (Fig. 3), whereas biodistribution may be different in patients with solid tumors or other cancer types, such as a retroperitoneal tumor with thyroid uptake (Fig. 4) or cutaneous lymphoma (Fig. 5), respectively. In addition to visual inspection of the images, biodistribution also may be assessed by evaluating the TBRT (day 6 or 7). For the tositumomab and 131I-tositumomab therapeutic regimen, the expected biodistribution is defined as a TBRT of between 50 and 150 h. snmjournals
Essential Role of Nuclear Medicine Technology in Tositumomab and 131I-Tositumomab Therapeutic Regimen for Non-Hodgkin's Lymphoma, William C. Cole1, Jennifer Barrickman, CNMT2 and Glen Bloodworth, CNMT2
How long does it take for Bexxar to work?
When given as initial treatment: "Responses were observed in 72 of the 76 patients, most of whom reported regression of palpable tumor within two weeks. Complete responses were observed in 57 of 76 patients (Table 1), with a median time to an evaluated complete response of 202 days (range, 55 to 693). The five-year rate of progression-free survival for all patients was estimated at 59 percent (95 percent confidence interval, 49 to 71) (Fig. 2).
The median progression-free survival was 6.1 years (95 percent confidence interval, 3.0 years to [upper confidence level not reached]), with a median follow up of 5.1 years. The 5-year progression-free survival for patients with a complete response was 77 percent (95 percent confidence interval, 67 to 89); 40 of the 57 patients (70 percent) who had a complete response (53 percent of the entire study population) remained in complete remission for 4.3 to 7.7 years after treatment."
~ NEJM, February 3, 2005 Vol. 352 no. 5 -131 I-Tositumomab Therapy as Initial Treatment for Follicular Lymphoma
What about repeated use of Bexxar?
"There are very limited data on repeated dosing with radioimmunoconjugates. Can it be done? In Kaminski's recent paper in Blood, seven of the 53 patients got a second dose in order to get a better response, but it didn't work. Only one patient went from stable disease to partial response. None of the other patients improved the quality of their response. However, there were 16 patients who progressed after getting the antibody and were re-treated. They had nine responses of the 16, including five complete remissions, with a median progression-free survival of 11 and a half months. So, yes, they can be re-administered." ~ Dr Bruce Cheson - bloodline.net
Repeat treatment with iodine-131-rituximab is safe and effective in patients with relapsed indolent B-cell non-Hodgkin's lymphoma who had previously responded to iodine-131-rituximab
annonc.oxfordjournals.org
Where to receive treatment with Bexxar
BEXXAR Treatment Center Locations
For the most up-to-date list of Treatment Centers,
contact the BEXXAR Service Center at 1-877-4BEXXAR (1-877-423-9927).
Centers that Administer Bexxar by State (last update, 2007)
What about toxicity to the bone marrow?
Relationship of degree of bone marrow involvement with hematologic toxicity in patients with non-Hodgkin's lymphoma treated with tositumomab and iodine I 131 tositumomab. ASCO 2003
Lack of treatment-related MDS/AML in patients with follicular lymphoma after frontline therapy with tositumomab and iodine I 131 tositumomab ASCO 2003
The iodine I-131 tositumomab therapeutic regimen: Summary of safety in 995 patients with relapsed/refractory low grade (LG) and transformed LG non-Hodgkin's lymphoma (NHL) jco.org
|
"Two studies have evaluated the risk for myelodysplastic syndrome (MDS) and acute myeloid leukemia following RIT with either 90Y-ibritumomab or 131I-tositumomab. These retrospective analyses of large numbers of patients both showed that RIT did not produce a higher risk for MDS in comparison with similar patient populations treated with multiple chemotherapies alone [90, 91]. MDS has rarely been observed in patients treated upfront with RIT using 131I-tositumomab alone [63].
- Radiolabeled and Native Antibodies and the Prospect of Cure of Follicular Lymphoma http://bit.ly/7mcys1
|
.gif) |
Specific Regimen Influences Risk of Myelodysplasia After Lymphoma Treatment BloodJournal
Aug 2005: "... none of the 76 patients with previously untreated, advanced-stage follicular
large granular non-Hodgkin lymphoma (LG-NHL) who received [bexxar] as initial therapy
developed t-MDS or t-AML over the 4.6 years of median follow-up.
"While longer follow-up is needed," the authors conclude, "these results suggest that
radioimmunotherapy will, with acceptable toxicity, take its place among the more effective
therapies for LG-NHL."
|
.gif) |
Harvesting Stem Cells for Transplant in Non-Hodgkin's Lymphoma Patients
Is Still Possible After Treatment with Bexxar news.cornell.edu
What Dr. Shore found was that, in 13 of 16 patients undergoing a clinical trial of Bexxar at NewYork-
Presbyterian Hospital’s Weill Cornell Medical Center, a harvesting of stem cells from the blood
produced enough stem cells for these patients to have a transplant. “The prior use of Bexxar does
not preclude adequate stem cell collection for autologous [self-donated] stem cell transplantation
in non-Hodgkin’s lymphoma,” Dr. Shore said. “This allows the patients who relapse after Bexxar
the option to proceed to transplant, whereas, before, it was uncertain if this could ever be done.”
|
Hematological (blood cell) Toxicity
in Patients receiving Bexxar as Initial Treatment:
| Variable |
Absolute Neutrophil Count |
Hemoglobin |
Platelet Count |
Median (average)
nadir (lowest) value |
1300 per mm3 |
12.2 g/dl |
83,000 per mm3 |
| Median time to nadir |
47 days |
44 days |
29 days |
| Toxicity (%) * |
|
|
|
| Grade 3 or 4 |
34 of 76 pts |
0 |
17 of 76 pts |
| Grade 4 |
5 of 76 pts |
0 |
0 |
Median duration
of toxicity** |
|
|
|
| Grade 3 or 4 |
22 days |
NA |
22 days |
| Grade 4 |
5 days |
NA |
NA |
|
Median time to return to baseline grade
|
60 days |
56 days |
43 days |
* Common Toxicity Criteria of the National Cancer Institute define a grade 3 absolute Neutrophil count as 500 to less than 1000 per cubic millimeter and a grade 4 count as less than 500 per cubic millimeter, a grade 3 hemoglobin level as 6.5 to less than 8.0 g per deciliter and a grade 4 level as less than 6.5 g per deciliter, and a grade 3 platelet count as 10,000 to less than 50,000 per cubic millimeter and a grade 4 count as less than 10,000 per cubic millimeter.
** The duration of grade 3 or 4 toxicity was defined as the number of days from the last count before grade 3 or 4 toxicity to the first day of the documented return to grade 2 toxicity.
Adapted from 131 I-Tositumomab Therapy (Bexxar) as Initial Treatment for Follicular Lymphoma ~ Kaminiski et al, NEJM, Feb 3 2005.
Aug 2005: "...none of the 76 patients with previously untreated, advanced-stage follicular large granular non-Hodgkin lymphoma (LG-NHL) who received tositumomab and I131 tositumomab as initial therapy developed t-MDS or t-AML over the 4.6 years of median follow-up.
"While longer follow-up is needed," the authors conclude, "these results suggest that radioimmunotherapy will, with acceptable toxicity, take its place among the more effective therapies for LG-NHL." - Medscape Specific Regimen Influences Risk of Myelodysplasia After Lymphoma Treatment -
HAMA
About one-third of patients with relapsed B-cell malignancies develop human
anti-mouse antibody (HAMA) following mouse antibody treatment.
.gif) |
See also HAMA
|
.gif) |
Survival benefit associated with human anti-mouse antibody (HAMA) in patients with
B-cell malignancies. Cancer Immunol Immunother. 2006 Dec;55(12):1451-8.
Epub 2006 Feb 22. PMID: 16496145
Patients with B-cell malignancies that developed high HAMA titers had longer survival
that was not explained by risk factors or histologic grade, suggesting the importance of the
immune system.
|
"Although approximately 10% of patients treated with tositumomab and iodine I 131 tositumomab (bexxar) developed human-anti-mouse antibodies, treatment with tositumomab does not preclude the
administration of subsequent chimeric (mouse-human) antibody therapies."
A clinical and scientific overview of tositumomab and iodine I 131 tositumomab.
Semin Oncol. 2003 Apr;30(2 Suppl 4):22-30. Review. PMID: 12728404
Potential contraindications for use
.gif) |
Bone marrow biopsy results:
.gif) |
Hypocellular bone marrow (<15% cellularity)
|
.gif) |
Marked reduction of bone marrow precursors
NOTE: The bone marrow findings must be within 30 days
|
|
.gif) |
Impaired bone marrow reserves as indicted by:
.gif) |
prior myeloablative therapies with ABM or PBSC transplantation
|
.gif) |
Platelet count < 100,000 cells/mm3
|
.gif) |
ANC (Neutrophil Count (Absolute)) < 1,500 cells/mm3
|
.gif) |
History of failed stem cell collection
|
|
.gif) |
Known type I hypersensitivity or anaphylactic reactions to murine (mouse) proteins
or to any component of the therapeutic regimen
|
.gif) |
Positive pregnancy test - test required for women of child-bearing age.
|
.gif) |
Serum Creatinine > 1.5 X the upper limit of normal
|
.gif) |
Previous external beam radiation involving >25% of the active bone marrow
|
.gif) |
Pregnancy and continuing breast feeding
|
.gif) |
Children and adolescents under 18 years of age
|
.gif) |
Prior bone marrow or stem cell transplantation (this is being studied)
|
Adapted from: interactive.snm.org | eanm.org pdf
.gif) |
Re-Treatment With I-131 Tositumomab in Patients With Non-Hodgkin's Lymphoma
Who Had Previously Responded to I-131 Tositumomab ASCO
|
Does spleen involvement preclude use of radioimmunotherapy?
Residual splenomegaly in a patient who has otherwise successfully responded in other sites following chemotherapy for lymphoma is another reason for performing a splenectomy. In these cases, the procedure may be performed for both diagnostic and therapeutic reasons; it can determine if the splenomegaly is due to persistent lymphoma, and should this be true, it can potentially eliminate the focus of residual disease. A less common indication for splenectomy that may be seen more frequently in the future is to allow patients to become eligible for enrollment onto novel treatment protocols. An example of this is in patients with lymphoma refractory to conventional chemotherapy who were treated with radioimmunotherapy using a radiolabelled anti-CD20 antibody.
In some of these patients, splenomegaly was found to complicate treatment, as the large organ served as an “antigen sink”, effectively decreasing the dose of radionuclide available to treat other sites of disease. Thus, pretreatment splenectomy may be indicated to eliminate this complicating factor.
Basic outline for patients, including general precautions and safety information
BEXXAR is administered in two steps: (1) the dosimetric and (2) therapeutic steps.
Non-radioactive antibody (similar to Rituxan) is given before both the “dosimetric” infusion and the “therapeutic” infusion to improve distribution of these doses throughout the body - clear the blood of normal b-cells, so the more active radio-labeled dose is focused on tumor cells.
-
dosimetric dose: Following the cold antibody, "a trace amount of radio-labeled antibody, is given to evaluate the clearance of radiation from the patient’s body with gamma camera scans. This allows for patient-specific dosing of the therapeutic dose.
-
therapeutic dose: 7-14 days after the dosimetric step, the therapeutic dose is administered, again after administration of cold (unlabeled) antibody.
Following the therapy:
.gif) |
Avoid contact with infants, young children, and pregnant women.
|
.gif) |
Sleep in a separate bed separated by a distance of at least 6 feet (2 to 3 meters).
|
.gif) |
Maintain an appropriate distance of 6 feet (2 meters) from others.
|
.gif) |
Travel alone in a private automobile if possible, otherwise maintain as great a distance as possible
between patient and driver.
|
.gif) |
Toilet should be used instead of urinal. Sit on toilet to prevent splashing, and flush several times after use.
|
.gif) |
Separate laundry and eating utensils and wash items separately.
|
"The amount of radiation received by close contacts of patients who followed the instructions is well within the guidelines deemed acceptable by the government agency regulating radiation exposure, namely, the Nuclear
Regulatory Commission (NRC)." See also Facts on Bexxar PDF
Ask your Nuclear Medicine physician for more specific information, which can vary by State.
Information & Resources:
.gif) |
About Bexxar: cancerbacup.org | bexxar.com | Oliver Press MD, PhD
|
.gif) |
Complete Prescribing Information for BEXXAR®
(Tositumomab and Iodine I 131 Tositumomab) us.gsk.com pdf
|
.gif) |
Bexxar: Characteristics, Clinical Pharmacology and Rationale for Dosing Regimen http://www.nucmedtutorials.com/dwrit-nhl/bexxar3.html
|
Background articles
.gif) |
Facts on Bexxar PDF (Dr. Oliver Press, LRF)
|
.gif) |
Bexxar®: Novel Radioimmunotherapy for the Treatment of Low-Grade and Transformed
Low-Grade Non-Hodgkin’s Lymphoma ~ The Oncologist, Vol. 9, No. 2, 160–172,
April 2004 theoncologist.alphamedpress.org
|
.gif) |
Articles on Bexxar drugs.com
|
.gif) |
Overview of Radioimmunotherapy: Clinical Experience With Tositumomab,
Iodine I-131 Tositumomab Mark S. Kaminski, MD - Introduction:
Why Radioimmunotherapy? medscape (free login)
|
.gif) |
Patient-Specific Dosing: The Critical Role of Dosimetry
Richard L. Wahl, MD - Dosimetry in RIT Medscape (free login)
|
.gif) |
Tositumomab, Iodine I-131 Tositumomab as Front-Line Therapy in Non-Hodgkin's Lymphoma
John Leonard, MD Radioimmunotherapy for NHL Medscape (free login)
|
.gif) |
Future Directions With Iodine-131 Tositumomab in Non-Hodgkin's Lymphoma
Oliver W. Press, MD, PhD Tositumomab: What the Future Holds Medscape (free login)
|
.gif) |
FDA Drug Advisory transcripts on Bexxar PDF
|
.gif) |
Radioimmunotherapy Agents: What They Are and How They Work bloodline.net
|
.gif) |
Subsequent therapy can be administered after tositumomab and iodine I-131 tositumomab
for non-Hodgkin lymphoma. Cancer. 2005 Dec 16; PMID: 16362977
|
Related Topics
.gif) |
|
.gif) |
HAMA and pet mice?
|
Related Abstracts
.gif) |
NEW: Re-Treatment With I-131 Tositumomab in Patients With NHL Who Had Previously Responded to I-131 Tositumomab http://jco.ascopubs.org/cgi/content/full/23/31/7985
In summary, the results of the current study suggest that re-treatment with I-131 tositumomab may be a potentially valuable therapeutic choice for patients who responded previously to therapy with I-131 tositumomab. Re-treatment with I-131 tositumomab appears to be relatively safe and effective and can result in durable responses in a subset of patients. Given these encouraging results, further studies of re-treatment with I-131 tositumomab have been designed and will include patients who prospectively enroll onto new clinical studies of I-131 tositumomab and have a duration of response of at least 6 months.
|
.gif) |
Combo Therapy with Tositumomab (Bexxar) & autoSCT Aids Elderly Lymphoma Patients cancerpage.com
"These data suggest that potentially curative therapies should not be denied to patients based solely on age," lead investigator Dr. Ajay K. Gopal of the University of Washington, Seattle, told Reuters Health. "In addition, targeted therapies with individualized dosing strategies, as in this study, may prove useful to both reduce toxicity and improve outcomes in patients of all ages."
|
.gif) |
The impact of FLIPI on outcome of frontline treatment with single-agent I-131 tositumomab for follicular lymphoma ASCO 7509 - 2006
|
.gif) |
New treatment, CHOP followed by Bexxar, shows long-term remission in patients with follicular non-Hodgkin’s lymphoma bexxar s0016.pdf
“We feel that the five-year results of the trial are tremendously encouraging and some of the best ever observed in a SWOG clinical trial for patients with advanced follicular non-Hodgkin’s lymphoma,” said Dr. Press, who is a member of the Fred Hutchinson Cancer Research Center, professor of medicine at the University of Washington and chairman of the scientific advisory board of the Lymphoma Research Foundation.
|
.gif) |
Iodine-131-Rituximab Radioimmunotherapy [Bexxar] in Patients With Relapsed or Refractory Indolent Non-Hodgkin's Lymphoma: Australian Multicenter Phase II Clinical Study. J Clin Oncol. 2006 Aug 28; PMID: 16940276
The objective overall response rate (ORR) was 76%, with 53% attaining a complete response (CR) or CR unconfirmed (CRu). Median duration of response for patients achieving CR/CRu was 20 v 7 months for those with a partial response (P = .0121). Median progression-free survival for the entire cohort was 13 months, with 14% remaining relapse free beyond 4 years. Median follow-up was 23 months, with a 4-year actuarial survival rate of 59% +/- 10%. Toxicity was principally hematologic; grade 4 thrombocytopenia occurred in 4% and neutropenia occurred in 16% of patients, with nadirs at 6 to 7 weeks after treatment.
|
.gif) |
Bexxar Following Fludarabine Produced Response in 100% of patients [first line] med.cornell.edu Sep 2005
|
.gif) |
Health Canada approves new targeted radioimmunotherapy ~ Bexxar(TM) Receives Approval to Treat Follicular Non-Hodgkin's Lymphoma newswire.ca
|
.gif) |
Radioactive Anti-CD 20 Antibody (Bexxar®) May Improve Outcome of Autologous Transplants for Follicular Lymphomas cancerconsultants.com
|
.gif) |
Efficacy and Safety of Tositumomab and Iodine-131 Tositumomab (Bexxar) in B-Cell Lymphoma, Progressive After Rituximab. J Clin Oncol. 2004 Dec 21; PMID: 15613695
|
.gif) |
Tositumomab and Iodine I 131 Tositumomab [Bexxar] for Recurrent Indolent and Transformed B-Cell Non-Hodgkin's Lymphoma. J Clin Oncol. 2004 Apr 15;22(8):1469-79. PMID: 15084620 | Related articles
|
.gif) |
ASH 2003 - [89] The BEXXAR Therapeutic Regimen (Tositumomab and Iodine I 131 Tositumomab) Produced Durable Complete Remissions in Heavily Pretreated Patients with Non-Hodgkins Lymphoma (NHL), Rituximab-Relapsed/Refractory Disease, and Rituximab-Naive Disease. abstractsview.com
|
.gif) |
Techniques for using bexxar for treatment tech.snmjournals.org.pdf
Describes pre-dosing; how dosing is calculated (with dosimetic dose); why the need: individual biologic clearance, etc. For professionals, but readable. Also tells about importance of avoiding infusion leaks.
|
.gif) |
High-dose radioimmunotherapy (Bexxar) versus conventional high-dose therapy and autologous hematopoietic stem cell transplantation for relapsed follicular non-Hodgkin's lymphoma: a multivariable cohort analysis. Blood. 2003 May 15 PMID: 12750161 PubMed
|
.gif) |
ASCO 2003 - Bexxar analysis of long-term responses prnewswire
|
.gif) |
Bexxar 2
Bexxar has a radioactive substance called iodine 131 attached to it. The monoclonal antibody in Tositumomab (Bexxar) targets a protein (CD20) found on the surface of mature B cells and the radioactive iodine delivers radiation directly to these cells. This destroys the lymphoma B cells. Unfortunately it may also affect some normal cells. (FDA is considering approval)
|
.gif) |
ASH 2002: Durable Responses With Bexxar (Tositumomab) In Transformed Non Hodgkin's Lymphoma Docguide.com
|
.gif) |
ASH: Durable Responses Reported with Single Dose of Tositumomab and Iodine I-131 Tositumomab (Bexxar) in Relapsed, Refractory Low-grade Non-Hodgkin's Lymphoma Docguide.com Dec_9_02
|
.gif) |
Efficacy & Background: Antibody Therapy for Non-Hodgkin's Lymphoma - Mark S. Kaminski, M.D. Professor of Internal Medicine Director, Multidisciplinary Lymphoma Clinic details
|
.gif) |
Efficacy as Single Agent in Utreated Patients: Iodine-131: Effective Front-Line Follicular Lymphoma Treatment -- Of the 76 pts with previously untreated advanced state follicular lymphoma, 74 had either a complete response (76%) or a partial response (21%). Progression-free survival was markedly better for the patients who had a complete response than for those who had a partial response. As estimated by Kaplan-Meier survival curves based on current data, the median progression-free survival for all patients who had a response was projected to be 59% at three years. Survival curves projected a 71% progression-free survival at three years for pts who had a complete response. ASCO
|
.gif) |
Efficacy: BEXXAR EFFECTIVE AS FIRST-LINE TREATMENT FOR NON-HODGKIN'S LYMPHOMA PATIENTS WHEN USED WITH CHEMOTHERAPY detail
|
.gif) |
Efficacy: Bexxar (Tositumomab and Iodine (I)-131-Tositumomab) Plus CHOP Improves Best Result in Newly Diagnosed Follicular Non-Hodgkin's Lymphoma. details
|
.gif) |
Efficacy: Complete response rate was increased 5-fold by addition of Bexxar, compared to fludarabine alone. details
|
.gif) |
Efficacy: Integrated Analyses of 251 Patients Treated with Bexxar Shows Thirty-Five Percent of Patients Achieved Durable Complete Responses Lasting a Median of More Than Three Years details
|
.gif) |
Commentary: Dr. Bruce Cheson, bloodline
|
.gif) |
Patient Anecdote: Bruce's Bexxar Experience details
|
|