Survey responders: 139
Median Age 56 (range 29 - 77 years)
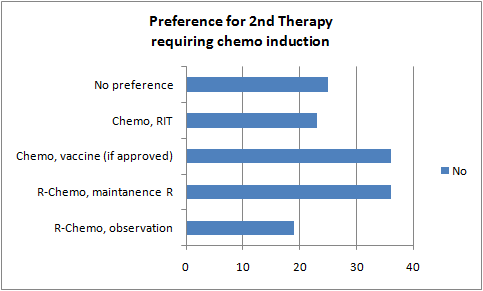
Preferred 2nd Treatment for follicular lymphoma (if chemo required)
| Preferred 2nd Treatment |
No. |
% |
| R-Chemo, observation |
19 |
14% |
| R-Chemo, maintenance R |
36 |
26% |
| Chemo, vaccine (if approved) |
36 |
26% |
| Chemo, RIT |
23 |
17% |
| No preference |
25 |
18% |
| Confidence in preference |
No. |
% |
| Convinced |
18 |
13% |
| Confident |
66 |
47% |
| Moderate confidence |
45 |
32% |
| Low confidence |
10 |
7% |
Beware of confidence! ... blessed are the unsure, because they will continue to ask questions.
| Would Consider Trials |
No. |
% |
| Only if Onc discusses |
60 |
43% |
| Even If Onc Doesn't |
72 |
52% |
| Never |
7 |
5% |
Limitations: The online community is a young "crowd" (median 56 years) and may have attitudes that do not predict those in the general population, which is ~10 years older. Further, the sample was non-random - which can lead to biased results ... for example, those with No Preference may be less likely to participate in the survey. Further there is no way to be certain that people will correctly self-identify their preferences and attitudes.
Discussion: As we know, preferences are not equivalent to what is best - that will require comparative trials with long follow up to determine. We decided to leave out plain "vanilla" RIT and Rituxan (without chemo induction) to make the choices more comparable, anticipating that almost everyone would choose Rituxan if it was among the choices (in order to avoid the chemo). We note further that the individual's unique clinical circumstances can be the critical factor in the selection of therapy.
The survey was prompted by an investigator's request who hoped to establish that vaccine could compete with Rituxan as a consolidation option (assuming it is approved - and we truly have no opinion or expectation if it will prove to be effective or not). This is the reason that only one investigational choice was provided.
What the preference data suggests is that if chemo is received as induction therapy that consolidation of some type is preferred to observation (to waiting for it to come back) ... in this group of patients, noting the above limitations.
Regarding vaccines: We expect that if vaccine was proven equivalent to Rituxan maintenance, in terms of efficacy (such as time to progression), the side effect profile would significantly favor vaccine ... so the high preference for vaccine (36%) was expected ... and we further expect that those favoring it over Rituxan would increase when presented with the relative safety data (again, assuming equivalence in other respects).
A caveat of vaccine therapy is that it requires a surgical procedure (a biopsy) to produce it. If this is a minor issue, we can't say.
However, crystal clarity is not the rule in clinical science - more typically we have to deal with a tradeoff - studies showing one protocol better in potential efficacy, but worse in potential toxicity. ... The answer requiring follow up sufficient to see a survival advantage for one versus the other protocol in randomly selected groups of patients. Importantly, correlative studies are needed to help predict individual outcomes - to identify biomarkers that predict which individuals will benefit from which protocol, and what should be avoided.
Finally, until such time, we can expect that the opinion of our doctors will influence what is prescribed (probably significantly)... maybe not so much with this confident group, but almost certainly in the general population.
Many, many thanks to the participants!
Karl S (PAL)