Topic Search: PubMed
A goal of clinical research is to deliver the right drug protocol to the right patient - based on the right diagnosis of the disease - the underlying biology.
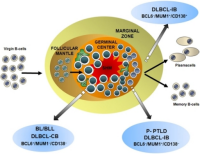 For DLBCL progress has been made ... but we have gotten ahead of ourselves. Routine immunohistochemistry (IHC) tests on biopsy samples do not appear to be reproducible (reliable) and therefore are not yet useful for guiding the approach to initial therapy for DLBCL. Further, we are not yet at a point where a reliable finding of cell of origin (COO) can guide the approach to therapy. For DLBCL progress has been made ... but we have gotten ahead of ourselves. Routine immunohistochemistry (IHC) tests on biopsy samples do not appear to be reproducible (reliable) and therefore are not yet useful for guiding the approach to initial therapy for DLBCL. Further, we are not yet at a point where a reliable finding of cell of origin (COO) can guide the approach to therapy.
Within, Perry et al. describe the limitations of IHC tests relative to tests using Gene Expression Profiling (GEP) on snap frozen fresh tissue ... and the need for a reliable test that is practical to use in the clinical setting.
The second report describes a promising new assay that could help to select patients for clinical trials based on the cell of origin and treatment agents that may be more active in different cell types.
Related Reports
.gif) |
Snip from:
Biological Prognostic Markers in Diffuse Large B-Cell Lymphoma
Anamarija M. Perry, MD, Zdravko Mitrovic, MD, and Wing C. Chan, MD
"Numerous biological prognostic markers have been proposed in patients with diffuse large B-cell lymphoma (DLBCL), and the significance of many of these that were studied before the rituximab era need to be reassessed. Prognostic markers are assayed by a variety of methods, most commonly by morphology and immunohistochemistry.
Although widely available and relatively cheap, the immunohistochemistry method suffers from poor reproducibility and difficulty in quantification due to differences in tissue processing, staining protocols, and inter-observer variability.
Molecular methods such as gene expression profiling (GEP), high-resolution array comparative genomic hybridization, and next-generation sequencing hold great promise in elucidating the pathogenesis and prognosis of DLBCL, but these methods are not widely available due to substantial cost, technical complexity, and requirement for fresh and frozen tissue. However, more focused assays can be designed for further studies, and the ability to apply these assays to formalin-fixed, paraffin-embedded tissue would allow the inclusion of large patient cohorts to improve statistical power.
Next-generation sequencing has detected numerous mutations in patients with DLBCL, but whether any of those mutations are important for prognosis, alone or in combination, is not yet answered. However, such global studies allow us to examine markers in the context of other modifying factors and hence overcome the problems of single-marker studies. For example, BCL2 may be an important biomarker, but its clinical significance is influenced by other factors such as other biological activities of the pathway that leads to its expression, the coexisting factors such as MYC translocation, and possibly even mutations that may affect its biological activities. Thus, studying BCL2 expression alone as a biomarker may not generate reproducible results from different populations.
A more global approach may allow all of these factors to be included in the analysis, thereby producing a more meaningful biomarker profile.
At the present time, there is no consensus on which biological prognostic markers should be routinely assessed in patients with DLBCL, and practices vary widely among different institutions. With more global approaches, such as those noted above, the ability to assess biomarkers in the cellular or tumor context may be possible, resulting in a better understanding of their biological and prognostic significance."
A reliable test for cell of origin in DLBCL appears to be on it's way:
|
.gif) |
ASH 2013: Determining Cell-Of-Origin Subtypes In DLB Lymphoma Using Gene Expression Profiling On Formalin-Fixed Paraffin-Embedded Tissue – An L.L.M.P.P. Project http://bit.ly/T3XrNA
The diffuse large B-cell lymphoma (DLBCL) cell-of-origin (COO) distinction into germinal center B cell (GCB) and activated B cell (ABC) subtypes, as molecularly described by our group, has profound biological, prognostic, and potential therapeutic implications.
New therapeutic agents with selective activity in ABC and GCB DLBCL are under development. An accurate diagnostic assay is urgently needed to qualify patients for clinical trials using targeted agents and as a predictive biomarker.
Although the subtypes were originally defined using gene expression profiling on snap-frozen tissues (frozen-GEP), it has become common practice to use less precise but relatively inexpensive and broadly applicable immunohistochemical (IHC) methods in formalin-fixed paraffin-embedded tissue (FFPET).
We sought to create a robust, highly accurate molecular assay for COO distinction using new GEP techniques applicable to FFPET. This new assay shows excellent performance in archival FFPET, and the rapid turn-around time (<36 hours from FFPET block to result) will allow prospective implementation in future therapeutic trials and, ultimately, clinical practice.
|
.gif) |
Oncologist, 2012:
Are We Ready To Stratify Treatment for Diffuse Large B-Cell Lymphoma Using Molecular Hallmarks? http://1.usa.gov/1pFaMt5
Current treatment paradigms do not distinguish between COO phenotype, and this can be readily justified. Several large analyses have shown no difference in prognosis between phenotypes using IHC classification methods, indicating a lack of precision and reliability that dissuades against routine clinical use.
GEP has superior accuracy in classifying DLBCL into COO subtypes with different biology and prognosis. It holds promise for future clinical application once issues of availability, cost, preference for analyses to be performed on fresh frozen tissue, lack of standardization in COO phenotype classification methods, and lack of prospective randomized clinical evidence validating its use can be overcome.
|
|